Total number of atoms in 44 g of Carbon dioxide is

Greenhouse gas - Wikipedia

PPT - EMPIRICAL FORMULA PowerPoint Presentation, free download - ID:662576

How many atoms are present in 44 gm of CO2? - Quora

SOLVED: A 11.0 g sample of carbon dioxide (molar mass 44.0 g/mol). How many molecules does it contain? A. 6.02x10^23 B. 3.01x10^23 C. 1.50x10^23 D. 2.40x10^24 2. What is the mass of

Which has the maximum number of molecules among the following?(a) 44 g CO2 (b) 48 g O3(c) 8 g H2
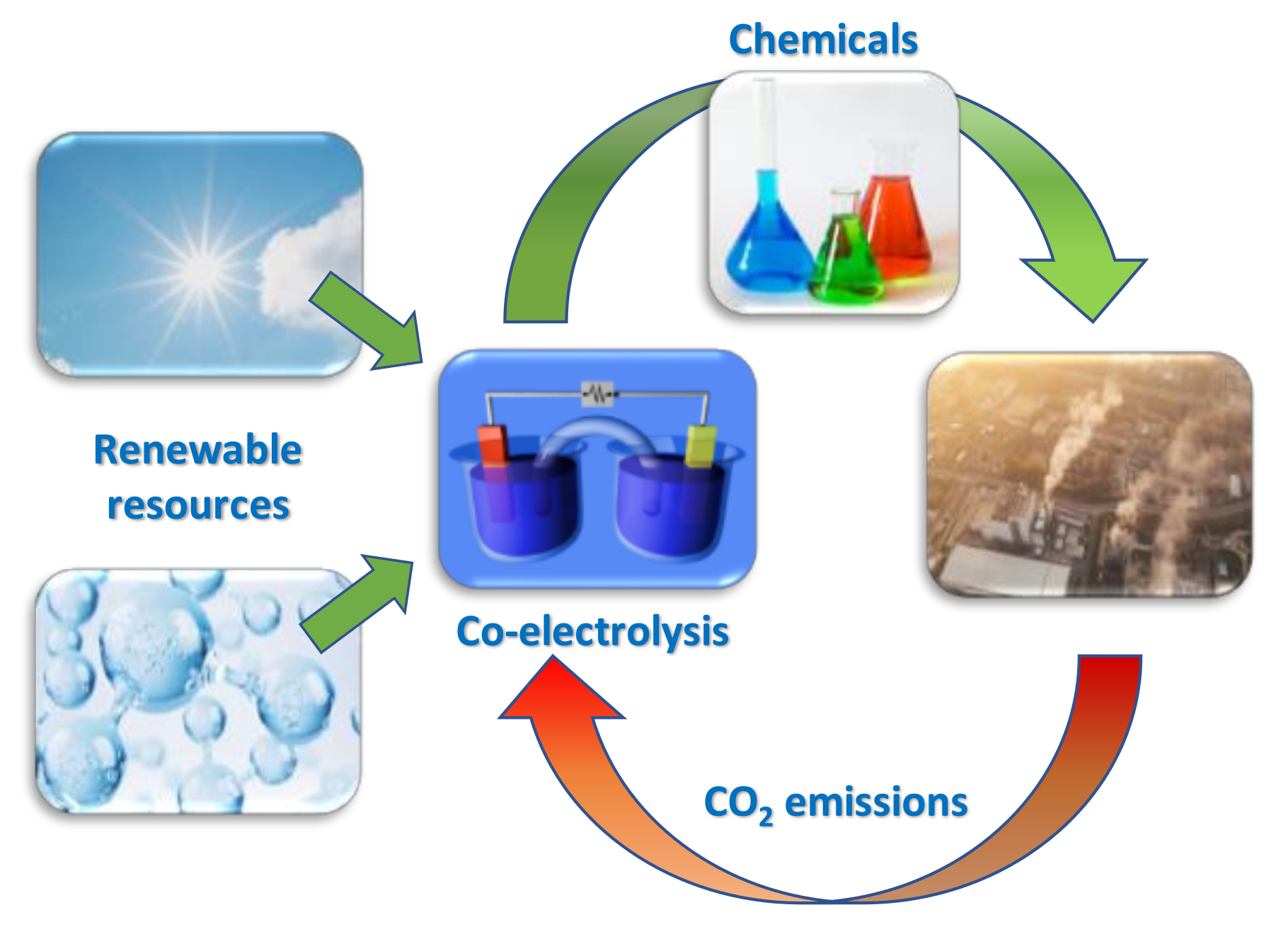
Catalysts, Free Full-Text
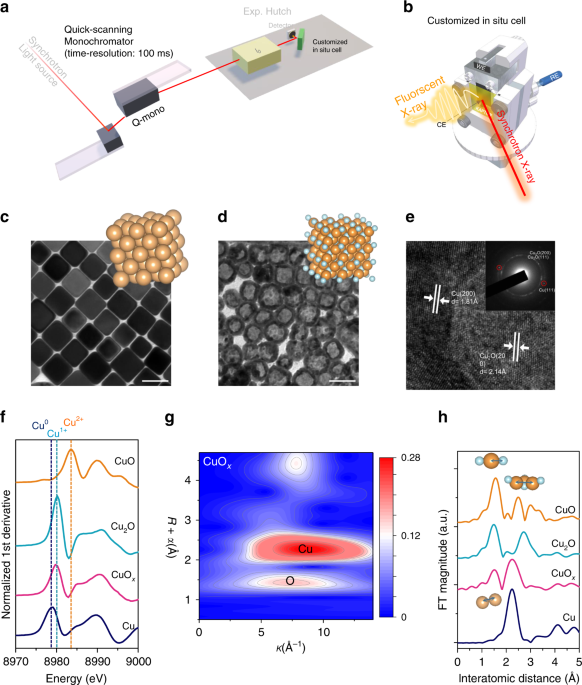
Operando time-resolved X-ray absorption spectroscopy reveals the chemical nature enabling highly selective CO2 reduction

Carbon - Wikipedia
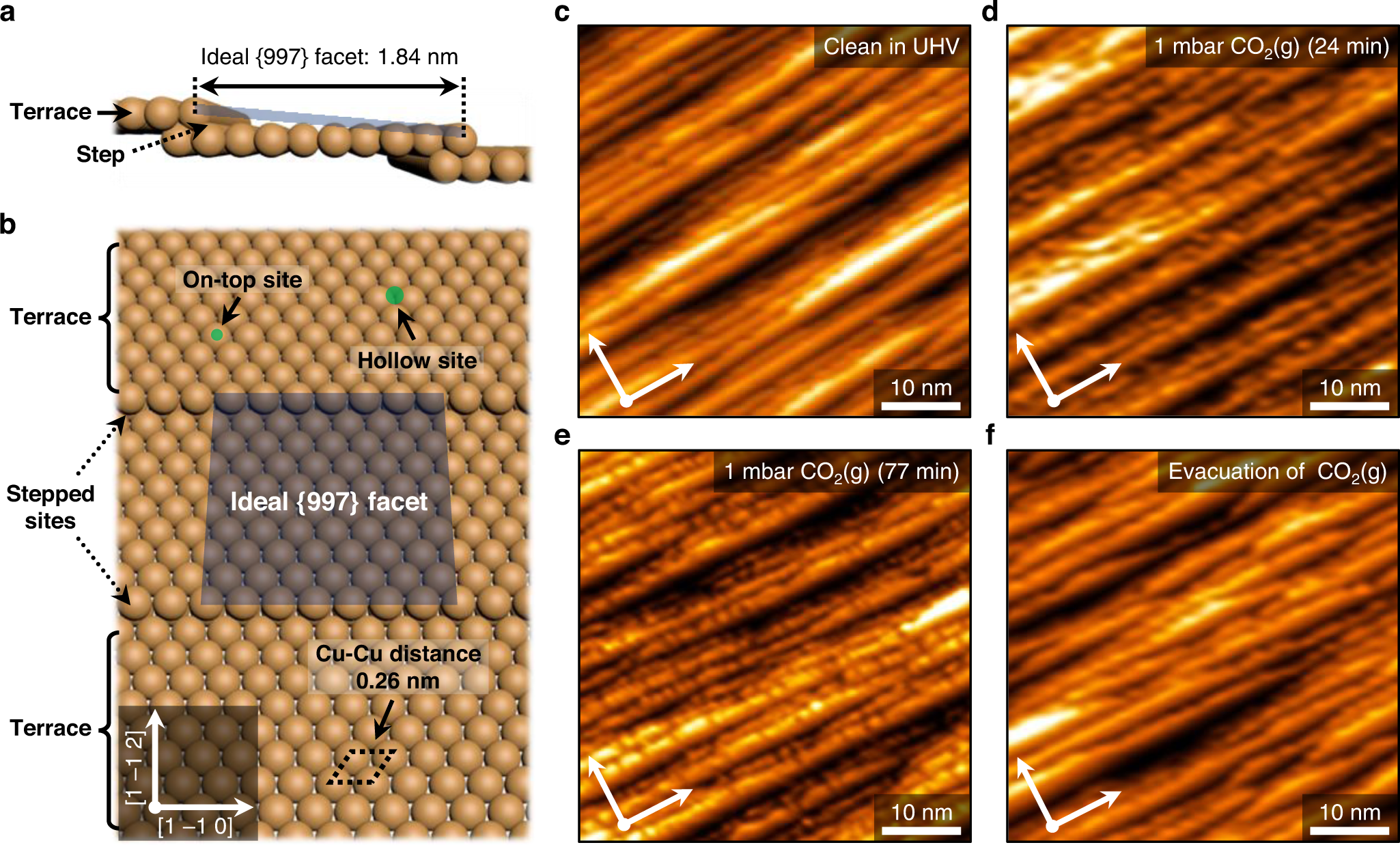
Revealing CO2 dissociation pathways at vicinal copper (997) interfaces
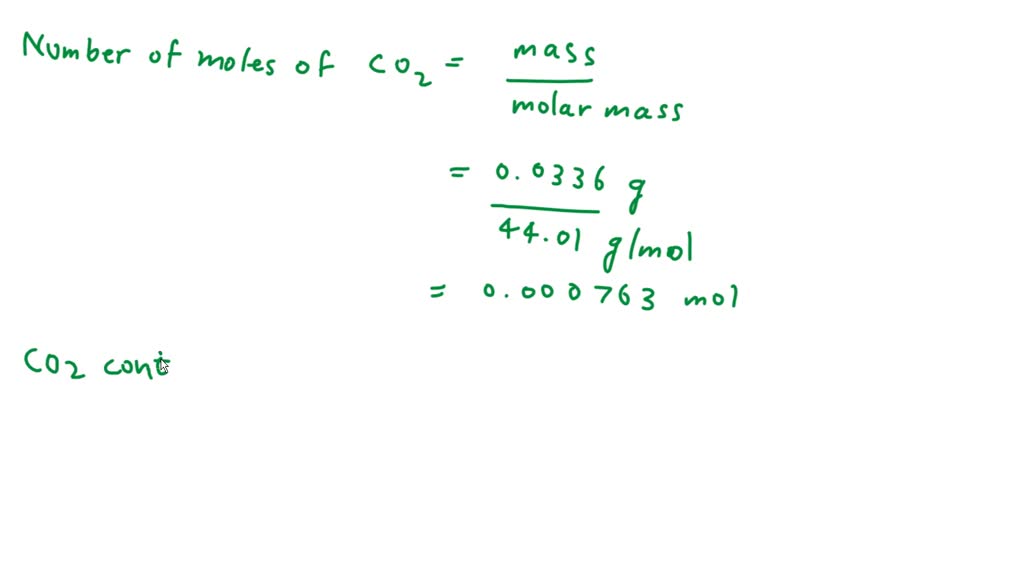
SOLVED: Atoms in 0.0336 g of CO2. The molar mass of CO2 is 44.01 g/mol. Find the number of moles of oxygen.